Описание
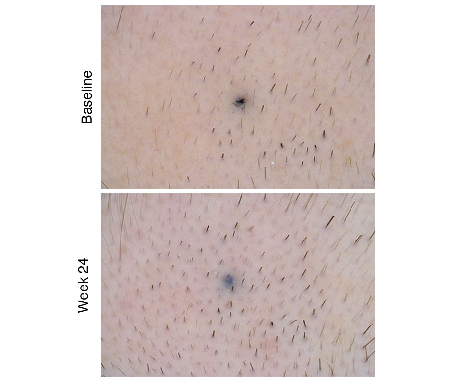
The finasteride 0.25% topical spray applicator and its mode of administration on the scalp are depicted. Developed to minimize systemic exposure compared with oral finasteride, the spray formulation targets hair follicles directly for treatment of male androgenetic alopecia.
Figure 1
Photograph
454 × 304px
· 78,0 КБ
Source Paper
Efficacy and safety of topical finasteride spray solution for male androgenetic alopecia: a phase III, randomized, controlled clinical trial.Journal of the European Academy of Dermatology and Venereology : JEADV (2022)
PMID: 34634163
DOI: 10.1111/jdv.17738
Cite This Figure
 > Source: B M Piraccini et al. "Efficacy and safety of topical finasteride spray solution for male androgenetic ." *Journal of the European Academy of Dermatology and Venereology : JEADV*, 2022. PMID: [34634163](https://pubmed.ncbi.nlm.nih.gov/34634163/)
<figure> <img src="https://pdfs.citedhealth.com/figures/34634163/118.png" alt="The finasteride 0.25% topical spray applicator and its mode of administration on the scalp are depicted. Developed to minimize systemic exposure compared with oral finasteride, the spray formulation targets hair follicles directly for treatment of male androgenetic alopecia." /> <figcaption>Figure 1. The finasteride 0.25% topical spray applicator and its mode of administration on the scalp are depicted. Developed to minimize systemic exposure compared with oral finasteride, the spray formulation targets hair follicles directly for treatment of male androgenetic alopecia.<br> Source: B M Piraccini et al. "Efficacy and safety of topical finasteride spray solution for male androgenetic ." <em>Journal of the European Academy of Dermatology and Venereology : JEADV</em>, 2022. PMID: <a href="https://pubmed.ncbi.nlm.nih.gov/34634163/">34634163</a></figcaption> </figure>