Dietary Clostridium butyricum and 25-Hydroxyvitamin D3 modulate bone metabolism of broilers through the gut-brain axis.
Study Design
- Study Type
- Other
- Population
- Rat model
- Intervention
- Dietary Clostridium butyricum and 25-Hydroxyvitamin D3 modulate bone metabolism of broilers through the gut-brain axis. 75 mg
- Comparator
- None
- Primary Outcome
- Body weight
- Effect Direction
- Neutral
- Risk of Bias
- Unclear
Abstract
Leg disorders have become increasingly common in broilers, leading to lower meat quality and major economic losses. This study evaluated the effects of dietary supplementation with Clostridium butyricum (C. butyricum) and 25-hydroxyvitamin D3 (25-OH-D3) on bone development by comparing growth performance, tibial parameters, Ca and P contents of tibial ash, bone development-related indicators' level, and cecal short-chain fatty acids in Cobb broilers. All birds were divided into four treatment groups, which birds fed either a basal diet (Con), basal diet + 75 mg chlortetracycline/kg (Anti), basal diet + C. butyricum at 109 CFU/kg (Cb), basal diet + C. butyricum at 109 CFU/kg and 25-OH-D3 at 25 μg/kg (CbD), or basal diet + 25-OH-D3 at 25 μg/kg (CD). Our results suggest that the dietary supplementation in Cb, CbD, and CD significantly increased the body weight (BW) and average daily gain (ADG), and reduced the feed-to-weight ratio (F/G) at different stages of growth (P < 0.05). Dietary supplementation in Cb, CbD, and CD prolonged (P < 0.05) the behavioral responses latency-to-lie (LTL) time, reduced (P < 0.05) the levels of osteocalcin (BGP) and peptide tyrosine (PYY), and increased (P < 0.05) serotonin (5-HT) and dopamine (DA). Treatment with Cb increased (P < 0.05) the levels of acetic acid, isobutyric acid, butyric acid, and isovaleric acid compared with those in Con group. The cecal metagenome showed that Alistipes spp. were significantly more abundant in Cb, CbD, and CD groups (P < 0.05). A total of 12 metabolic pathways were significantly affected by supplementation, including the signaling pathways of glucagon, insulin, and PI3K-AKT; primary and secondary bile acid biosynthesis; and P-type Ca 2+ transporters (P < 0.05). Hence, the CbD supplementation modulates bone metabolism by regulating the mediators of gut-brain axis, which may inform strategies to prevent leg diseases and improve meat quality in broilers.
Full Text
Dietary Clostridium butyricum and 25-Hydroxyvitamin D3 modulate bone metabolism of broilers through the gut—brain axis
Guangtian Cao,* Yang Yu,y Huixian Wang,y Huijuan Yang,* Fei Tao,* Shenglan Yang,y Jinsong Liu,z Zhanming Li,x and Caimei Yang y,1
*College of Standardisation, China Jiliang University, Hangzhou 310018, PR China; yKey Laboratory of Applied Technology on Green-Eco-Healthy Animal Husbandry of Zhejiang Province, Zhejiang Provincial Engineering Laboratory for Animal Health and Internet Technology, College of Animal Science and Technology, Zhejiang A & F University, Hangzhou 311300, PR China; zZhejiang Vegamax Biotechnology Co., Ltd, Anji 313300, PR China; and xSchool of Grain Science and Technology, Jiangsu University of Science and Technology, Zhenjiang 212004, PR China
ABSTRACT Leg disorders have become increasingly common in broilers, leading to lower meat quality and major economic losses. This study evaluated the effects of dietary supplementation with Clostridium butyricum (C. butyricum) and 25-hydroxyvitamin D3 (25-OH-D3) on bone development by comparing growth performance, tibial parameters, Ca and P contents of tibial ash, bone development-related indicators’ level, and cecal short-chain fatty acids in Cobb broilers. All birds were divided into four treatment groups, which birds fed either a basal diet (Con), basal diet 75 mg chlortetracycline/kg (Anti), basal diet + C. butyricum at 109 CFU/kg (Cb), basal diet + C. butyricum at 109 CFU/kg and 25-OH-D3 at 25 mg/kg (CbD), or basal diet + 25OH-D3 at 25 mg/kg (CD). Our results suggest that the dietary supplementation in Cb, CbD, and CD significantly increased the body weight (BW) and average daily gain (ADG), and reduced the feed-toweight ratio (F/G) at different stages of growth
(P < 0.05). Dietary supplementation in Cb, CbD, and CD prolonged (P < 0.05) the behavioral responses latency-to-lie (LTL) time, reduced (P < 0.05) the levels of osteocalcin (BGP) and peptide tyrosine (PYY), and increased (P < 0.05) serotonin (5-HT) and dopamine (DA). Treatment with Cb increased (P < 0.05) the levels of acetic acid, isobutyric acid, butyric acid, and isovaleric acid compared with those in Con group. The cecal metagenome showed that Alistipes spp. were significantly more abundant in Cb, CbD, and CD groups (P < 0.05). A total of 12 metabolic pathways were significantly affected by supplementation, including the signaling pathways of glucagon, insulin, and PI3K-AKT; primary and secondary bile acid biosynthesis; and P-type Ca 2+ transporters (P < 0.05). Hence, the CbD supplementation modulates bone metabolism by regulating the mediators of gut−brain axis, which may inform strategies to prevent leg diseases and improve meat quality in broilers.
Key words: Clostridium butyricum, vitamin D, bone development, gut−brain axis, broiler
2024 Poultry Science 103:103966 https://doi.org/10.1016/j.psj.2024.103966
INTRODUCTION
Being one of the major sources of animal protein, demand for poultry has been increasing for decades. This has resulted in a drive to produce fast-growing, large broilers with good feed conversion rates, and high growth rates (Havenstein et al., 1994). The incubation temperature, transports time, feeding mode, body
2024 The Authors. Published by Elsevier Inc. on behalf of Poultry Science Association Inc. This is an open access article under the CC BY license (http://creativecommons.org/licenses/by/4.0/).
Received February 12, 2024. Accepted June 5, 2024. 1Corresponding author: [email protected]
weight, and growth rate of broilers can affect their leg strength, walking ability, and then meat quality (Knowles et al., 2008; Guz et al., 2020€ ). Under current practices, these factors combine to yield various negative health consequences, such as the development of contact dermatitis, arthritis, tenosynovitis, and claudication (Santos et al., 2022). Leg disorders are closely associated with poor growth and feed conversion, high mortality rate, carcass condemnation, and low meat quality (Oviedo-Rondon et al., 2009). In the USA, lameness and bone diseases in broilers cost manufacturers more than $150 million annually (Kieronczyk et al., 2017). Therefore, leg disorders in broilers during poultry production deserve more research attention.
1
Several studies have recently reported that the intestinal microbiota is closely related to bone health (Yan et al., 2016; Li et al., 2020). Specifically, changes in the intestinal microbiota can disrupt the normal functioning of the gastrointestinal tract and affect the growth and development of bones. Ohlsson et al. (2017) found that intestinal microorganisms activate the expression of receptor activator for nuclear factor-kb ligand (RANKL) and tumor necrosis factor-a (TNF-a) in mice, which induce the osteoclast formation and bone resorption. Furthermore, Mohammed et al. (2021) has confirmed the positive effects of probiotics on broilers’ bone development. Schwarzer et al. (2016) found that Lactobacillus plantarum supplementation promotes juvenile growth, and prevents developmental retardation in male CONV-R germ-free mice during chronic malnutrition. Oral treatment with Lactobacillus reuteri also improves dynamic measurements of the distal femur epiphysis, lumbar trabecular parameters, osteoblast serum markers, and bone formation in male mice (Mccabe et al., 2013).
Several factors involved in the gut−brain axisincluding intestinal microorganisms, short-chain fatty acids (SCFAs), and 5-hydroxytryptamine (5-HT)play important roles in regulating bone development. Yan et al. revealed that GF mice chronically colonized with microbiota show improved bone formation owing to the enhanced secretion of insulin-like growth factor 1 (IGF-1) (Yan et al., 2016). The gut microbiome also increases butyrate levels, which allows the parathyroid hormone (PTH) to boost Treg levels, thereby preventing bone loss (Li et al., 2020). The monoamine hormone and neurotransmitter, 5-HT, modulates bone remodeling and promotes bone formation (Lavoie et al., 2017). SCFAs not only prevent bone loss but also regulate the activity of bone cells and affect bone healing (Wallimann et al., 2021). Notably, C. butyricum has been shown to produce SCFAs, regulate the gut microbiota, and play a positive role in the gut−brain axis (Sun et al., 2021). In addition, commercially available vitamin D (25-OHD3) is an important feed additive that enhances bone development in broilers. However, it has some limitations in improving bone-related problems in birds during broiler production (Mitchell et al., 1997).
In a previous study, we showed that C. butyricum modulates the cecal microbiota, enhances butyrate production, and promotes bone development in broilers during the early stages of growth (Yu et al., 2022). Based on our findings, we hypothesized that C. butyricum regulates the gastrointestinal microbial structure of broilers and affects the secretion of hormones, brain −gut peptides, and neurotransmitters by changing the SCFA-mediated enteric-brain axis. Through this mechanism, C. butyricum may affect bone metabolism and prevent leg diseases in broilers. We also hypothesized that C. butyricum, when used in combination with 25-OHD3, may have an additive effect in mitigating skeletal problems. To test these hypotheses, we evaluated how C. butyricum (alone or in combination with 25-OH-D3)
affected the growth, intestinal flora, and bone metabolism of broilers.
MATERIALS AND METHODS Animal Management and Sample Collection
In total, 800 one-d-old male Cobb birds (obtained from Zhejiang Guangda Breeding Poultry Co., Ltd, Hangzhou, China) were randomized into 5 treatment groups (8 replicates, with 20 birds per replicate). The groups were fed the following diets: basal diet (Con), basal diet + 75 mg/kg chlortetracycline (Anti), basal diet + C. butyricum (CGMCC 9386) at 109 CFU/kg (Cb), basal diet + C. butyricum at 109 CFU/kg and 25OH-D3 at 25 mg/kg (CbD), and basal diet + 25-OH-D3 at 25 mg/kg (CD). In the building in which the birds were housed, the temperature was gradually reduced from 35 to 29°C at a rate of 2°C/wk. The feeding experiment was conducted for 42 d, feed and drinking water were provided ad libitum. Food intake and mortality were recorded daily. This study strictly observed the rules of the Ministry of Health of the People’s Republic of China, and the protocol was approved by the Animal Care and Use Committee of Zhejiang A&F University (ZAFUAC 2023043, Hangzhou, China). The basic nutritional levels and compositions of the diets followed NRC (1994) recommendations and are listed in Table 1. The probiotic and 25-OH-D3 used in the present study were provided by Zhejiang Vegamax Biotechnology Co., Ltd (Anji, China).
Table 1. Composition and nutrient levels of the basal experimental diet (air-dry basis).
Ages (days) 1−21 22−42
Items
Ingredients (%) Corn 56.33 57.4 Soybean meal 24.5 19 Extruded soybean 5 4 Distillers dried grains with solubles 5 8 Corn gluten meal 2 3 Soybean oil 1.2 4.3 Limestone 1.3 1.3 Fermented soybean meal 1.67 0 Premix1,2 3 3 Total 100.00 100.00
Nutrient levels Metabolizable energy (kcal/kg) 2949 3148 Crude protein (%) 20.6 18.6 Crude fat (%) 4.9 8.0 Lysine (%) 1.17 0.99 Methionine+Cysteine (%) 1.45 1.23 Threonine+Tryptophan (%) 1.13 0.95 Calcium (%) 0.88 0.79 Total phosphorus (%) 0.64 0.56 Available phosphorus (%) 0.39 0.34
- 1Minimal vitamin levels per kg of diets: vitamin A (retinyl acetate), 1,500 IU; vitamin D3 (cholecalciferol), 2,00 IU; vitamin E (DL a-tocopheryl acetate), 10 IU; riboflavin, 3.5 mg; pantothenic acid, 10 mg; niacin, 30 mg; cobalamin, 10 mg; choline chloride, 1,000 mg; biotin, 0.15 mg; folic acid, 0.5 mg; thiamine 1.5 mg; pyridoxine 3.0 mg.
- 2Minimal mineral levels per kg of diet: Fe, 80.00 mg; Cu, 8.00 mg; Mn, 60.00 mg; Zn, 40.00 mg; I, 0.18 mg; Se, 0.15 mg.
On d 42, one bird per replicate was selected for sample collection. Blood was sampled from the inferior wing vein of selected birds, following which the birds were peacefully euthanized. The blood samples were centrifuged at 3,000 £ g (25℃, 15 min) for serum collection, which serum was stored under -80°C until further use. Aseptic cryopreservation tubes and scissors were used to collect the cecal contents of birds. All cecal samples were stored at -80°C until SCFA analysis and microbial sequencing. The left tibia was manually defleshed, detaching the articular cartilage from the fresh bone, and then stored at -20°C, which all surrounding tissues were removed prior to examination of bone characteristics.
Growth Performance
Body weight was recorded on d 1, 21, and 42, and feed consumption was daily recorded, which determine the average daily feed intake (ADFI), average daily gain (ADG), and feed-to-body weight gain ratio (F/G).
Tibial Indices
The adherent muscles and other soft tissues were removed from the tibia, and the length, width, and weight of the bone were measured. Bone radio graphic density was measured as described in a previous study (Deng et al., 2010). The anteroposterior view was used to acquire radiographic images of the tibia (2.5 mAs; 45 kV). Following the methods used by Zhou et al. (2009), the bone index was calculated as: bone index (g/ kg) = tibial mass (g)/body mass at slaughter (kg). The length was determined from lateral intercondylar tubercle to inferior articular surface.
Bone-breaking strength was determined via a 3-point flexural bending test, following the methods outlined by Fleming et al. (Fleming et al., 1998). Each sample was kept at the same position, and held by cradle support. A 5kN load cell (20 mm/s speed) was imposed at the intermediate point, and the software Nexygen Plus (Lloyd Instruments, UK) was used for recording the bonebreaking strength (N) after suitable calibration.
Measurement of Ca and P Concentrations in Tibial Ash
The tibial samples were boiled, and all tissues and cartilage caps were removed as described in a previous study (Zhang, 2007). The Ca content of the bone was determined via the potassium ferrate method, and the P content was determined spectrophotometrically.
temperature of approximately 32°C. The latency was recorded as either the time until the birds squatted down and their body touched the water or 5 min.
Measurement of Cecal SCFA Levels
Cecal SCFA levels were measured using gas chromatography (GC) as described in a previous study (Cao et al., 2022), with minor changes. In short, 0.5 g of the cecal digesta from each bird was mixed with 1 mL of precooled double-distilled water and centrifuged at 10,000 £ g for 5 min at 4°C. The supernatant was then mixed with 25% metaphosphoric acid (v/v, 5:1), iced for approximately 0.5 h, and analyzed using an Agilent Technologies 7890B GC System (column dimensions: 30 m £ 0.25 mm £ 0.25 mm; Agilent Technologies, Palo Alto, CA).
Metagenomic Analysis of Cecal Contents
Metagenomic analysis of the cecal contents (3 birds per treatment group) was performed by Shanghai OE Biotech Co., Ltd. (Shanghai, China). Trimmomatic software (v. 2.0) was used to remove low-quality reads from the DNA data. Metagenomic sequences were compared in batches, and species abundance was calculated based on the results of comparative analysis. Predicted protein-coding genes were functionally annotated by comparing their sequences with those in the KEGG (https://www.kegg.jp/), eggNOG v5.0 (http://egg nog5.embl.de/#/app/home), CAZyme (http://www. cazy.org/), and other databases. Thus, we used the total metagenomic dataset to elucidate the functions of the microbiome. Finally, KEGG pathway analysis was used to screen and analyze pathways related to bone metabolism.
Measurement of Latency-to-Lie Time
Following the methods of Groves et al. (Groves and Muir, 2017), LTL was measured on d 37. Birds were placed in a 50 £ 35 £ 50 cm3 plastic basin containing water at a depth of approximately 3 cm and a
Statistical Analysis
IBM SPSS Statistics software (v. 26.0; IBM Corp., Armonk, NY) was used to analyze the data, and GraphPad Prism 8.0 (GraphPad Prism Inc., San Diego, CA) was used for data visualization. Measurements for each
treatment group were compared using an analysis of variance (ANOVA) and Duncan’s multiple range test and were expressed as the mean § the standard error of mean. Differences between groups were considered statistically significant at the 5% level (P < 0.05).
RESULTS Growth Performance
Tibial Parameters
Compared with birds in the Con and Anti groups, birds in the CD group had significantly longer tibias on average (P < 0.05; Table 2). Tibial length was significantly greater in the CD group than in the Con and Anti groups (P < 0.05). In addition, Cb and CD supplementation significantly increased tibial weight relative to the Anti group (P < 0.05). No significant inter-group differences were observed in the other tibial indices.
On d 21 and 42, the Anti, Cb, CbD, and CD supplementation significantly increased the BW of birds compared with Con treatment (P < 0.05, Table 2).
From d 1 to 21, ADG was significantly higher in the Anti, Cb, CbD, and CD groups than in the Con group (P < 0.05, Table 2); no significant difference was observed in ADG across these treatment groups. Moreover, CbD supplementation significantly increased the ADFI relative to the Con group (P < 0.05). F/G values were significantly lower in the Anti, Cb, and CD groups than in the Con, and CbD group (P < 0.05).
From d 22 to 42, ADG was significantly higher in the Anti, Cb, CbD, and CD supplementation groups than in the Con group (P < 0.05); F/G values were significantly lower in the Anti, Cb, CbD, and CD groups than in the Con group (P < 0.05). Moreover, the Cb, CbD, and CD treatments showed a significant improvement in ADFI relative to the Con group (P < 0.05). And, the CbD treatment significantly increased the ADG compare with Anti treatment (P < 0.05).
From d 1 to 42, Anti, Cb, CbD, and CD treatment significantly increased the ADG of birds, and decreased the F/G values compared with Con treatment (P < 0.05). And, the Cb, CbD, and CD treatments showed a significant increment in ADFI relative to the Con group (P < 0.05). And, the CbD treatment significantly increased the ADG and ADFI compare with Anti treatment (P < 0.05).
LTL, Tibial Ca and P Contents, and BMD and Bone-Breaking Strength
On average, LTL times were significantly longer for birds in the CbD and CD groups than for those in the Con, Anti and Cb groups (P < 0.05; Figure 1A). Moreover, the average LTL times for birds in the CbD and CD groups were significantly longer than those for Cbgroup birds (P < 0.05). The Cb group also had a significantly (P < 0.05) longer average LTL time than the Anti group. However, no significant inter-group differences were observed in the Ca and P contents of tibial ash (all P > 0.05; Figures 1B and 1C).
Compared with those in the Anti group, CbD-group birds had significantly higher tibial BMD of broilers (P < 0.05), and both CbD and CD-group birds had significantly (P < 0.05) higher bone-breaking strength (Figures 1D and 1E). The bone radiograph of all birds’ tibia was shown in Figure 1F.
Brain−Gut Peptides
Caecal levels of 5-HT and DA were significantly higher in Anti, and CD group birds than in Con- and CbD-group birds (P < 0.05; Figures 4A and 4B). No
significant i difference was found about the serum IGF-1 levels (P > 0.05; Figure 4C). Moreover, PYY levels were significantly higher in the CbD and Con group than in the Anti group (P < 0.05; Figure 4D). Hypothalamic 5HT and DA levels were significantly higher in the Anti, Cb, CbD, and CD groups than in the Con group (P < 0.05; Figures 4E and 4F).
SCFA Content of Cecal Digesta
Birds in the Cb group had significantly higher concentrations of acetic acid, isobutyric acid, butyric acid, and isovaleric acid in their cecal digesta than birds in the Con group (P < 0.05; Figures 5A, 5C−5E). Relative to the Con group, the Cb group had significantly lower levels of propionic acid (P < 0.05; Figure 5B). Levels of isobutyric acid and butyric acid were significantly higher in
the Cb group than in the Anti group (P < 0.05; Figures 5C and 5D).
Composition of the Gut Microbiota
In total, 261,988 genes were common across all samples; however, the Con and Anti groups had more unique genes than the other three groups (Figure 6A). The dilution curve indicated that with an increase in the number of samples extracted (on the abscissa), the number of core/pan genes extracted tended to reach saturation (on the ordinate; Figure 6B). In terms of community composition, the top 12 genera in all the treatment groups included Alistipes, Bacteroides, Helicobacter, Phocaeicola, Sutterella, Faecalibacterium, and Clostridium. Bacteria in the class Clostridia, Alistipes sp. An31A, Helicobacter pullorum, uncultured Bacteroides sp.,
Alistipes sp. Marseille-P5061, Bacillus spp., and bacteria in the family Rikenellaceae were the predominant species (Figure 6C). The abundance of Alistipes spp. at the genus level in Cb (17.2%), CbD (20.6%), and CD (20.6%) treatments’ birds were higher than those in the Con (13.8%) and Anti (11.2%). Moreover, the Cb group showed a significantly higher abundance of Bacteroides than the other groups. The Cb, CbD, and CD groups had a higher abundance of Alistipes sp. An31A than the Con and Anti groups. Furthermore, the abundance of Helicobacter pullorum was higher in Cb-treated birds than in the other groups (Figure 6D). PCA and PCoA analyses indicated that all samples from the Cb, CbD, and CD treatment groups were well separated from those collected from the Con and Anti treatment groups, suggesting that dietary supplementation in the Cb, CbD, and CD groups modulated the microfloral structure of broilers to a considerable degree (Figures 5E−5F).
The top 12 genus- and species-level taxa were selected for multi-factor functional difference analysis (Figures 7A and 7B). At the genus level, the Cb, CbD, and CD groups showed a significantly (P < 0.05) higher abundance of Alistipes than the Con and Anti groups. Moreover, the abundances of Azospirillum, Blautia, Desulfovibrio, Eubacterium, Faecalibacterium, Flavonifractor, and Pseudoflavonifractor were significantly lower in Cb and CD-group birds than in those in the Con group. The abundance of Parabacteroides was significantly low in the CbD group (P < 0.05). The abundances of Alistipes sp. An31A and Alistipes sp. Marseille-P5061 were significantly higher in the Cb and CbD groups than in the Con and Anti groups. The abundances of Bacteroides sp. CAG:714, Bacteroides uniformis, Parabacteroides distasonis, and Phocaeicola barnesiae were significantly low in the Cb and CbD groups. Moreover, the abundances of Alistipes sp. CAG:831 and Rikenellaceae spp. were significantly higher in the CD group than in the Con and Anti groups.
The results of LEfSe analysis showed that at the family level, Helicobacteraceae dominated the cecal microflora of birds in the Cb group; Eggerthellaceae, Porphyromonadaceae, Akkemansiaceae, and Prevotellaceae dominated in the CD group; Coriobacteriaceae, Flavobacteriales, and Campylobacteraceae dominated in the CbD group; and Streptococcaceae, Ruminococcaceae, Selenomonadales, Rhodospirillales, and Synergistaceae dominated in the Anti group (Figure 7C).
Functional analyses with PCA and PCoA revealed that all samples collected from the experimental groups (Cb, CbD, and CD) were well separated from those belonging to the Con and Anti groups (Figures 8A and 8B). Visualization with a heat map confirmed that scores for metabolic pathways including the glucagon signaling pathway, naphthalene degradation, primary and secondary bile acid biosynthesis, insulin signaling pathway, phenylpropanoid biosynthesis, PI3K-AKT signaling pathway, and chloroalkane and chloroalkene degradation were higher in the CbD group than in the Con and Anti groups and lower in the CD and Cb groups (Figure 8C). Multi-factor comparative analysis of the top 12 KEGG pathways (level 3) revealed that the PI3K-AKT signaling pathway, primary and secondary bile acid biosynthesis, insulin signaling pathway, phenylpropanoid biosynthesis, glucagon signaling pathway, naphthalene degradation, and chloroalkane and chloroalkene degradation were significantly promoted after dietary supplementation in the CbD group (Figure 8D).
DISCUSSION
Growth performance is the most basic and important indicator of broiler performance, and the positive effect of probiotics (when used as feed additives) on this measure broiler growth performance (Abdel-Hafeez et al., 2017). Studies have showed the growth promoting and immune enhancing effects of C. butyricum in broilers
and piglets (Liao et al. 2015; Takahashi et al., 2018; Cao et al., 2019). Our previous study also revealed that supplementation with C. butyricum in combination with 25-OH-D3 positively influenced the growth performance and bone metabolism of early broilers (Yu et al., 2022). The results of present study indicated that the Anti, Cb, CbD, and CD treatments significantly increased the BW and ADG compared with those in the Con treatmeng . In addition, the Con group had the highest F/G values among all groups. It is remarkable that the dietary
combination of C. butyricum and 25-OH-D3 increased the ADG and ADFI than Anti supplementation. These findings are in accordance with those of other studies showing the positive effects of probiotics on broilers (Mohammed et al., 2021; Santos et al., 2022).
Bone health affects the welfare of broilers, and maintaining good bone health in birds strongly aligns with the economic interests of breeders (Shim et al., 2012). Supplementation with 1,25(OH)2D3 has been shown to improve bone ash content in broilers and improve
cartilage dysplasia during different growth periods (Elliot et al., 1995). Similarly, supplementation with B. subtilis has been shown to significantly prolong LTL time, improve tibial length and weight, and improve the bone ash (Mohammed et al., 2021). Tibial bone mineral density (BMD) and ash content are susceptible to dietary Ca and P concentrations in broilers
(Liu et al., 2017). In the present study, we found that CbD supplementation increased tibial length and BMD, LTL, and bone strength in broilers. Moreover, supplementation with 25-OH-D3 alone (CD) or Cb improved the tibial weight of broilers. Rizzoli and Biver (2020) reported that probiotics improve the growth performance and strength of tibial bones by
increasing the digestibility of the diet and the resorption of Ca and P.
Serum levels of bone formation-related markers and hormones reflect the status of bone metabolism. Decreased levels of the propeptide of type I procollagen and osteocalcin have negative effects on bone structure and mineralization in broilers (Szulc et al., 2017). Moreover, gastrointestinal hormones are typically related to bone resorption-related markers and affect bone homeostasis (Jensen et al., 2021). Increased AKP activity is primarily related to increased osteoblastic activity in broilers, and can be treated as a positive marker to evaluate bone health (Asensio et al., 2020). The AKP activity of Cb, CbD and CD birds was higher than that of Anti birds, although wasn’t different from that of Con birds in present study. The BGP regulates bone resorption, and is positively in close contact with mineralization process, and bone turnover (Niimi et al., 2014). Other hormones such as IGF-1, PCT, and PTH have also been shown to affect bone development (Dixit et al., 2021). Among them, the PTH facilitates bone turnover and reduces the loss of bone (Silva et al., 2011). In the present study, the Cb, CbD, and CD treatment groups
had significantly lower levels of BGP than that of Con group, while higher than that of Anti group. In addition, the Anti, CB, and CD groups had significantly lower serum levels of PYY and PTH than the Con group; this indicated low levels of Ca in the blood, likely due to bone mineralization. Overall, the above results suggest that supplementation with C. butyricum and 25-OH-D3 changed the levels of bone formation-related markers and bone metabolism-related hormones, while more trials should be conducted to investigate the previous mechanisms.
Central 5-HT promotes bone development, prevents bone resorption, and increases bone mass in mouse (Yadav et al., 2009). Our data showed that the treatments with Cb, CbD, and CD increased the hypothalamic and cecal levels of 5-HT. In addition, the DA promotes bone remodeling and formation via the hypothalamic-pituitary-gonadal axis, and inhibits osteoclast differentiation via the cAMP/PKA/CREB pathway (Wang et al., 2021). Moreover, the Cb and CD treatments increased serum and intestinal levels of DA. Combined with the above studies, our data also suggest that the CbD treatment increased the production of 5-HT
and DA, thereby improving bone mass. And, its regulatory effects on DA were more significant than those on 5-HT.
SCFAs produced by intestinal microorganisms enhance the expression of tryptophan hydroxylase 1, which promotes the synthesis of 5-HT by intestinal chromaffin cells (Reigstad et al., 2015). SCFAs also promote the secretion of IGF-1, and regulate bone mass in mice (Yan et al., 2016). In the present study, dietary supplementation in the Cb group significantly increased the levels of the major SCFAs, which is inconsistent with the results of previous studies. Hence, we speculated that Cb regulates bone mass via the modulation of neural DA by SCFAs. The production of SCFAs is closely related to the changes in bacterial flora. Alistipes is the main producer of bacterial SCFAs, and Bacteroides produces propionic acid (De Vadder et al., 2016). Our metagenomic analysis indicated that Alistipes, Bacteroides, Faecalibacterium, and Clostridium were the dominant genera in all treatment groups, and that treatment with Cb significantly increased the abundances of these bacteria.
Previous studies on glucagon, insulin, and bone metabolism have indicated that the metabolism of bile acid by intestinal microbes regulates host immunity (Zhao et al., 2017), which this process is closely tied to classical bone metabolism via related signaling pathways (Cui et al., 2020). DA interacts with the adrenergic system to regulate glucagon and insulin secretion via adrenergic and dopaminergic receptors (Aslanoglou et al., 2021). Therefore, supplemental C. butyricum may affect bone metabolism by regulating the structure of the intestinal microflora, and this may be accompanied by the upregulation of various bone metabolism-related pathways. KEGG pathway analysis revealed that the abundance of P-type Ca2+ transporters differed from that typically associated with the signaling pathways of glucagon, insulin, PI3K-AKT, and primary and secondary bile acid biosynthesis. This explains the potential mechanism through which supplementation with C. butyricum upregulated the expression of bone formation-related markers, bone metabolism-related hormones, and SCFAs and confirms our initial hypothesis. In addition, Fox et al. (2008) suggested that P-type Ca2+ channels regulate the release of neurotransmitters, and these channels are selectively inhibited by 5-HT (Ciranna et al., 1996). Therefore, supplemental C. butyricum may regulate bone metabolism via the neurotransmitter 5-HT, although the specific mechanism of this effect needs to be further explored at the cellular and molecular level.
CONCLUSIONS
Our results suggest that supplementation with C. butyricum (at 109 CFU/kg) and 25-OH-D3 (at 25 mg/ kg) can improve growth performance of broilers by modulating the bone health and metabolism. Specifically, the combination of C. butyricum and 25-OH-D3
supplementation significantly increased the abundance of Alistipes and Bacteroidesis, the main strains producing SCFAs, and also modulated other mediators of the gut−brain axis (hormones and neurotransmitters). Our findings are of great importance for the broiler production industry, and may contribute to the development of novel management techniques to prevent leg diseases, thus reducing economic losses.
Figures
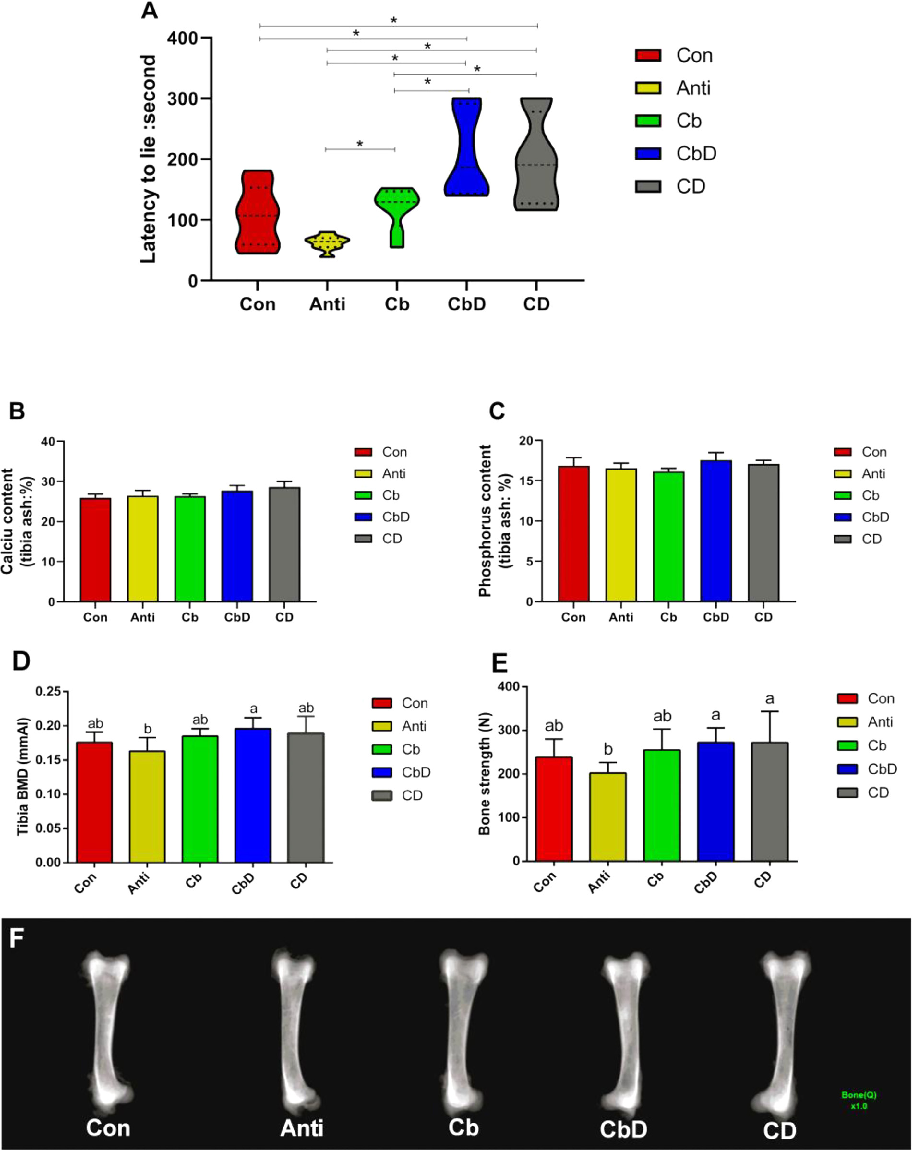
Figure 1
Effects of dietary Clostridium butyricum and 25-hydroxyvitamin D3 supplementation on latency-to-lie time in a poultry model, indicating improvements in leg health and bone strength.
chart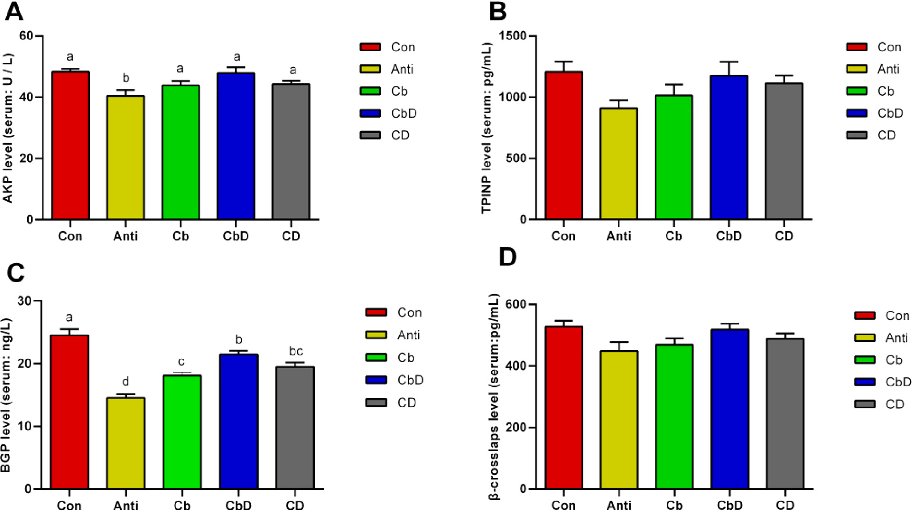
Figure 2
Experimental data from a study on dietary Clostridium butyricum and 25-hydroxyvitamin D3 and their combined effects on bone metabolism through gut microbiota modulation in poultry.
chart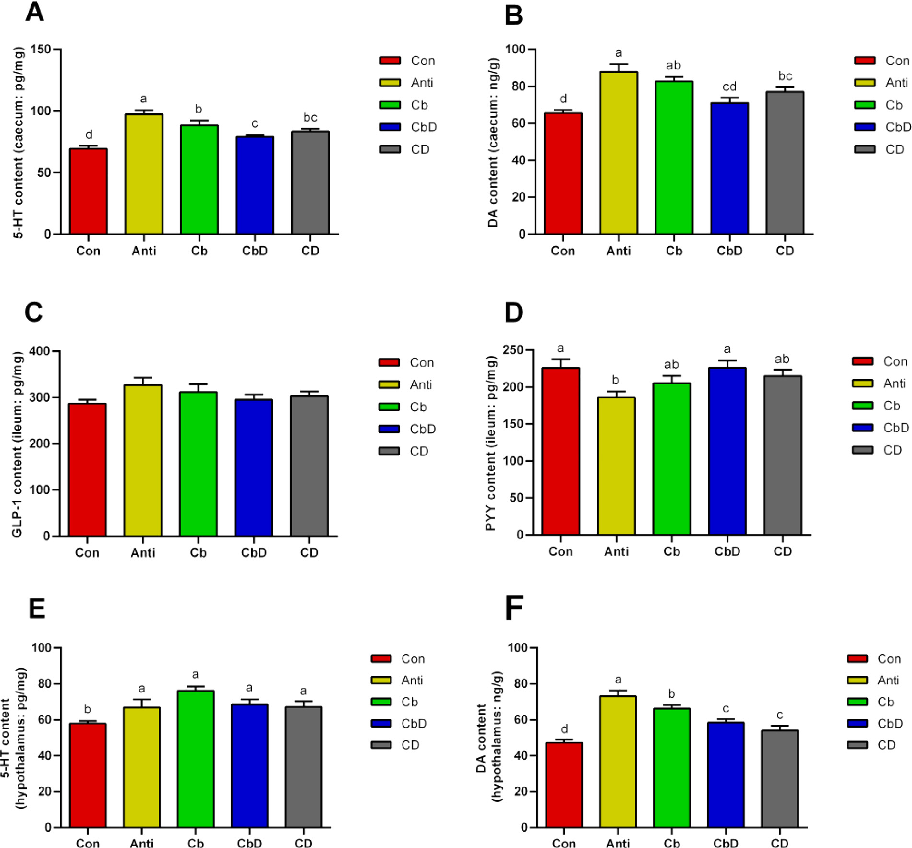
Figure 3
Hypothalamic signaling pathway analysis in poultry fed Clostridium butyricum and 25-hydroxyvitamin D3, examining effects on bone metabolism regulatory mechanisms.
chart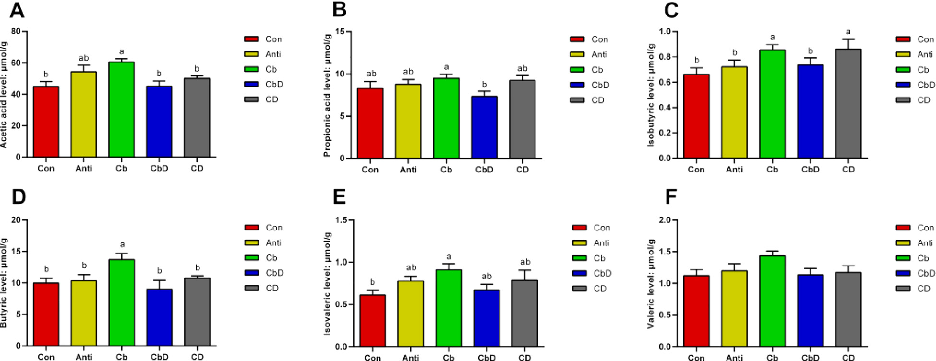
Figure 4
Caecal short-chain fatty acid (SCFA) concentrations in poultry supplemented with Clostridium butyricum and 25-hydroxyvitamin D3, linking gut fermentation products to bone metabolism modulation.
chart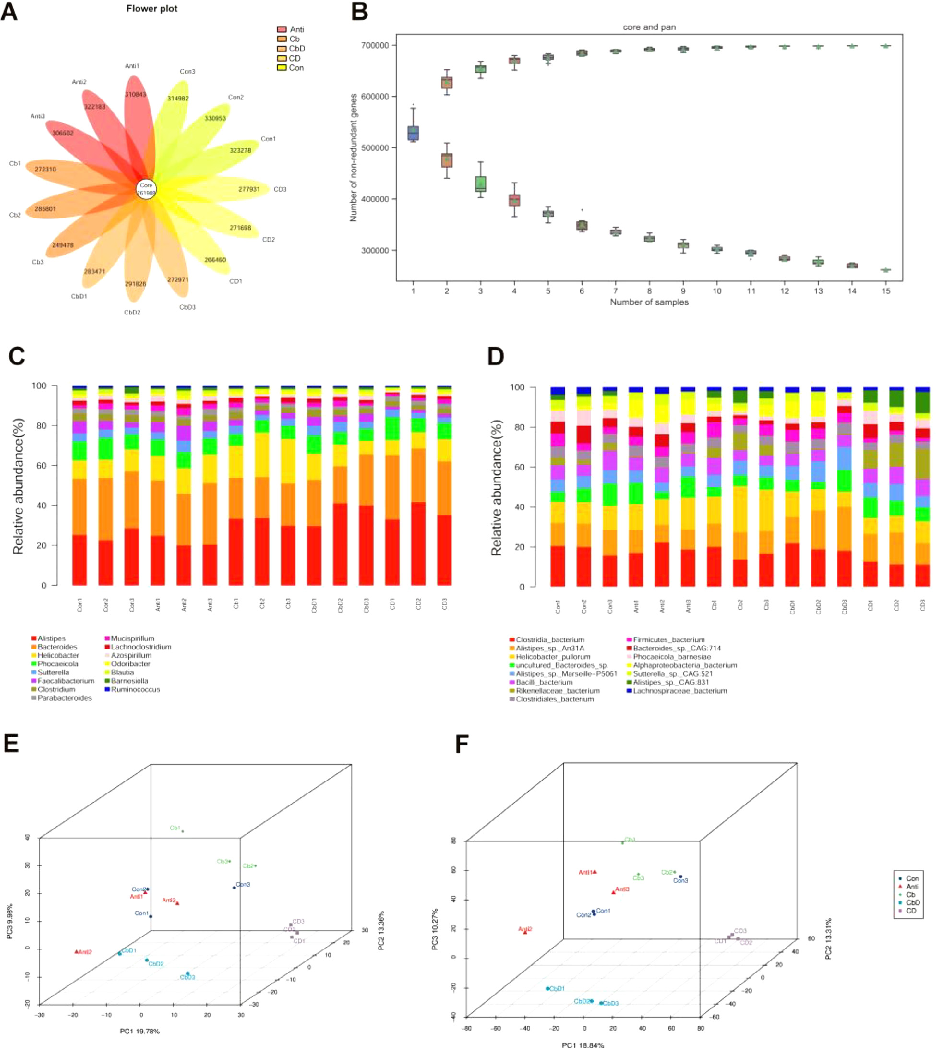
Figure 5
Metagenomic analysis of caecal microbiota in poultry receiving Clostridium butyricum and 25-hydroxyvitamin D3 supplementation, revealing shifts in microbial community structure.
chart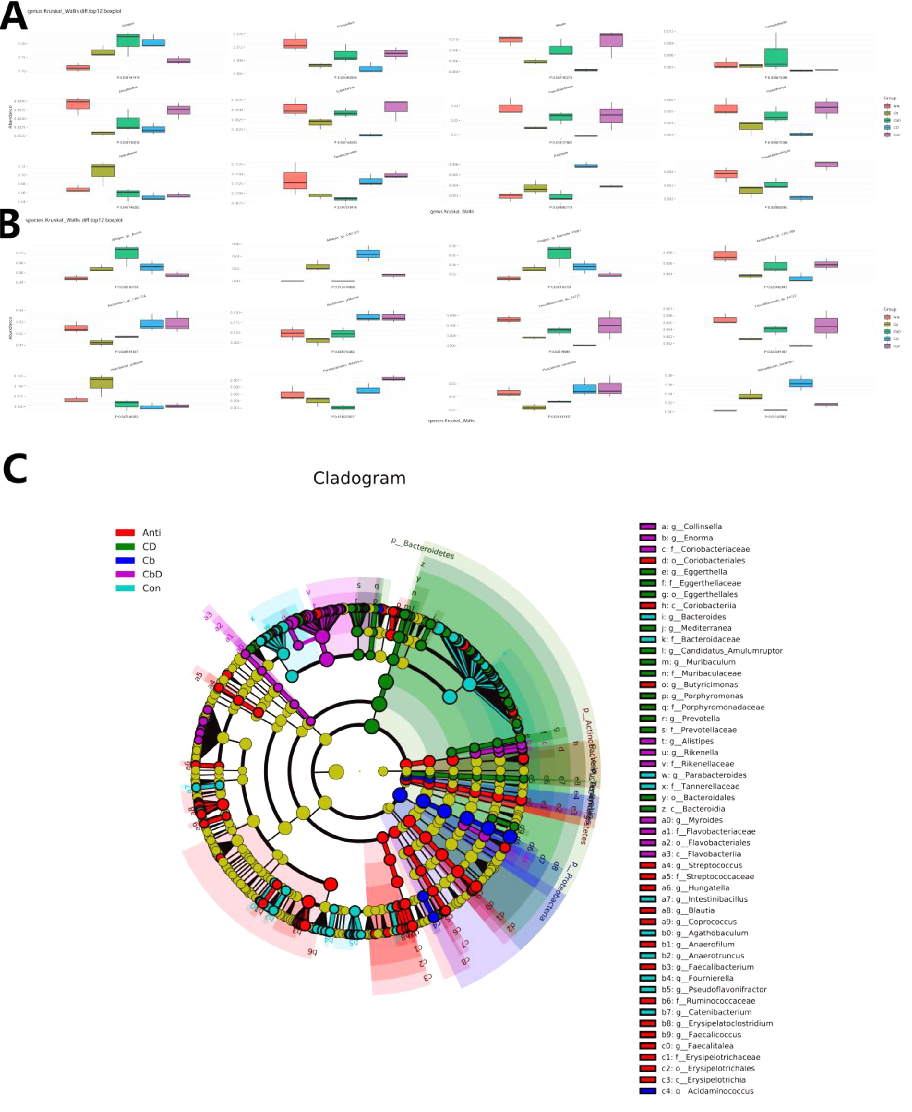
Figure 6
Caecal microbial composition data from poultry treated with Clostridium butyricum and 25-hydroxyvitamin D3, showing taxonomic-level changes associated with improved bone metabolism.
chart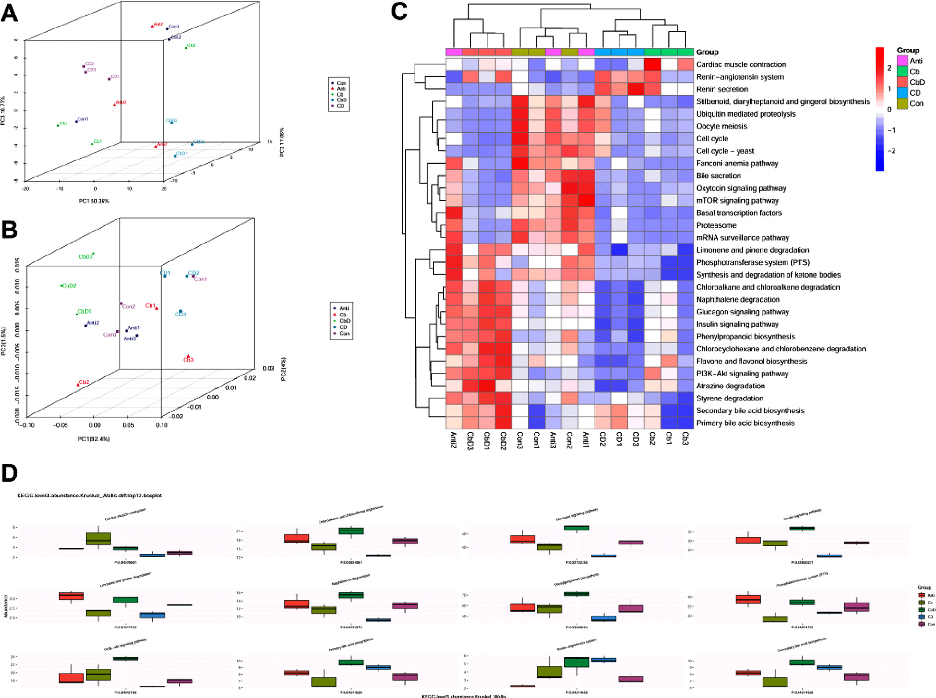
Figure 7
Metagenomic analysis of caecal microbiota in poultry receiving Clostridium butyricum and 25-hydroxyvitamin D3 supplementation, revealing shifts in microbial community structure.
chartUsed In Evidence Reviews
Similar Papers
Molecules (Basel, Switzerland) · 2019
Hepatotoxicity of a Cannabidiol-Rich Cannabis Extract in the Mouse Model.
Archives of biochemistry and biophysics · 2016
Zinc and skin biology.
Regenerative therapy · 2022
Regenerative medicine strategies for hair growth and regeneration: A narrative review of literature.
Indian journal of dermatology · 2017
Vitamin D Level in Alopecia Areata.
Journal of cosmetic dermatology · 2021
Role of vitamin D in hair loss: A short review.
Journal of cosmetic dermatology · 2016