Description
Sub-acute CBD dosing produced changes in liver weight and serum biochemistry parameters. This figure compiles multi-parameter hepatotoxicity data from the 10-day dosing study.
More Figures from This Paper
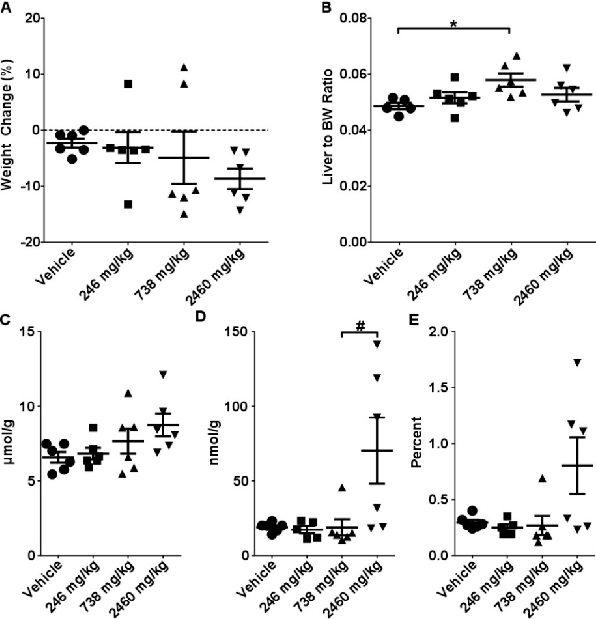
Figure 5
Single gavage with CBD at 246, 738, or 2460 mg/kg produced dose-dependent hepatotoxic effects in mice. Body weight changes, liver-to-body weight ratios, and intrahepatic total glutathione concentrations at 24 hours indicate significant liver burden at the highest doses.
chart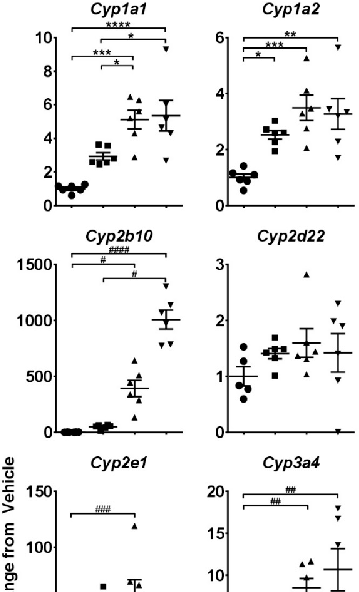
Figure 6
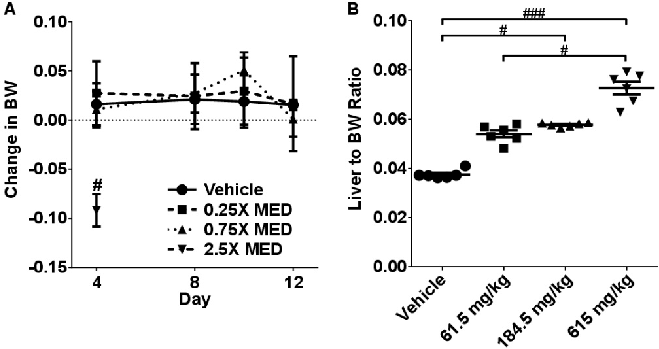
Sub-acute CBD administration (10 daily doses at 61.5-615 mg/kg) produced cumulative hepatic effects. This figure presents body weight curves and liver enzyme data from the two-week dosing protocol.
chart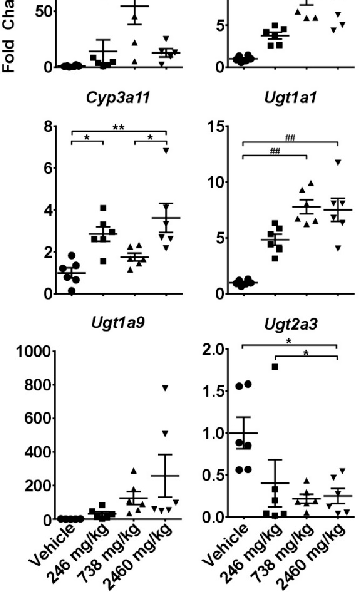
Figure 7
Single CBD gavage induced significant changes in hepatic cytochrome P450 and UDP-glucuronosyltransferase expression. Quantitative real-time PCR data demonstrate dose-dependent upregulation of these xenobiotic-metabolizing enzymes at 24 hours post-treatment.
chart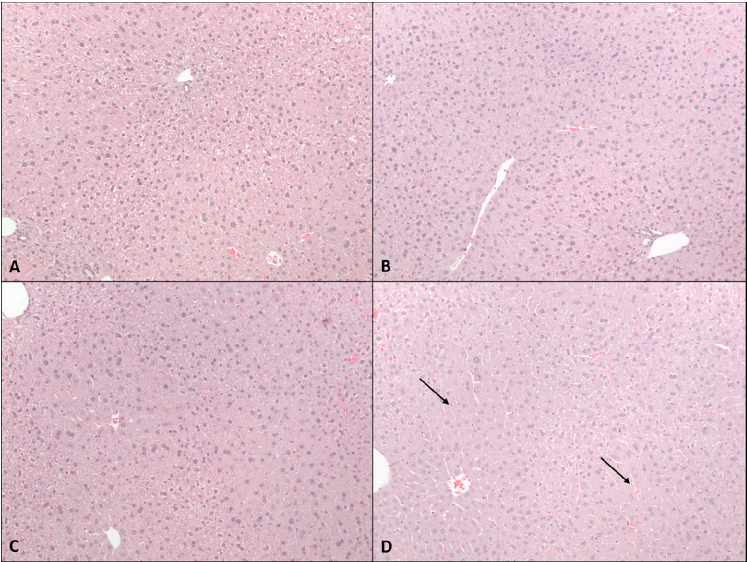
Figure 8
Hematoxylin and eosin-stained liver sections reveal dose-dependent histomorphological changes after 2-week CBD administration. Mice receiving 615 mg/kg CBD exhibited notable hepatocellular alterations compared to vehicle controls and lower-dose groups.
micrograph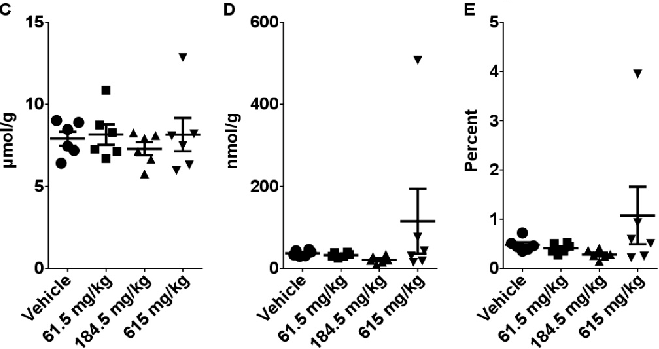
Figure 10
Hepatic gene expression profiling after sub-acute CBD treatment reveals broader metabolic impacts. This figure presents transcriptomic data on stress-response and metabolism-related genes in the liver.
chart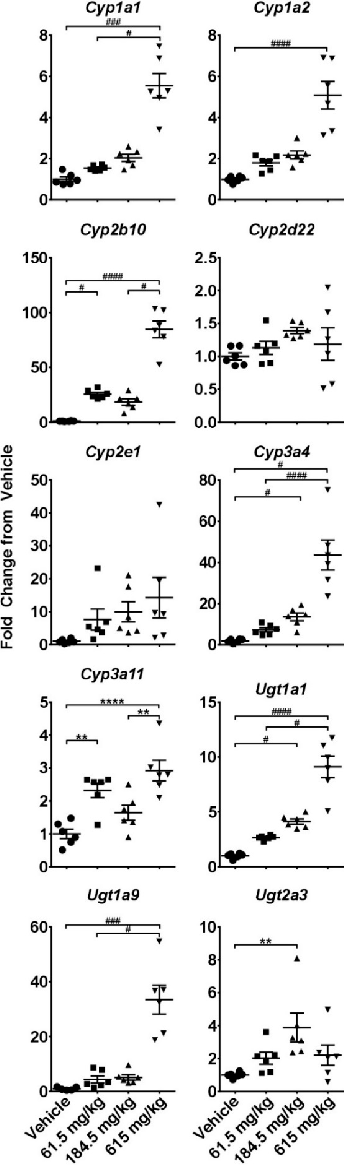
Figure 11
Two-week CBD administration produced sustained changes in cytochrome P450 and UDP-glucuronosyltransferase gene expression. Quantitative PCR data collected 6 hours after the last gavage indicate persistent hepatic enzyme induction at higher CBD doses.
chartFigure 9
ChartSource Paper
Hepatotoxicity of a Cannabidiol-Rich Cannabis Extract in the Mouse Model.Cite This Figure
 > Source: Laura E Ewing et al. "Hepatotoxicity of a Cannabidiol-Rich Cannabis Extract in the Mouse Model.." *Molecules (Basel, Switzerland)*, 2019. PMID: [31052254](https://pubmed.ncbi.nlm.nih.gov/31052254/)
<figure> <img src="https://pdfs.citedhealth.com/figures/31052254/119.png" alt="Sub-acute CBD dosing produced changes in liver weight and serum biochemistry parameters. This figure compiles multi-parameter hepatotoxicity data from the 10-day dosing study." /> <figcaption>Figure 9. Sub-acute CBD dosing produced changes in liver weight and serum biochemistry parameters. This figure compiles multi-parameter hepatotoxicity data from the 10-day dosing study.<br> Source: Laura E Ewing et al. "Hepatotoxicity of a Cannabidiol-Rich Cannabis Extract in the Mouse Model.." <em>Molecules (Basel, Switzerland)</em>, 2019. PMID: <a href="https://pubmed.ncbi.nlm.nih.gov/31052254/">31052254</a></figcaption> </figure>